
Each vial was dried by using a heat gun, cooled in a stream of argon, and placed in a glovebox before its use. Typical copolymerization experiments were performed in 2 mL vials. Thus, to insert both epoxide and anhydride moieties into the growing polymer chain, a pair consisting of a metal complex catalyst (Lewis acid) and a nucleophilic co-catalyst (Lewis base) has to co-operate. According to the proposed mechanism, molecules of epoxide must be first activated by its coordination to metal centers and then undergo the ring-opening reaction under the influence of the attack of free carboxylate ions. The newest experimental results supported by theoretical calculations point out that this stage occurs rather through a coordination insertion pathway than a concerted pathway proposed previously for epoxide/CO 2 copolymerization. The first-order dependence on epoxide concentration indicates that either epoxide binding or opening is the turnover-limiting step. 
The detailed mechanistic studies performed by the groups of Duchateau, Darensbourg, and Coates showed that the ring-opening of epoxide is the stage that determines the rate of ROCOP reactions in the presence of catalytic systems composed of metal complexes and nucleophilic co-catalysts. It was also shown that the properly designed catalytic systems based on Lewis acid–Lewis base pairs not only play a role of polymerization initiators by activating the molecules of epoxides but also influence on the rate of a propagation stage and the type of end groups in the final polyesters. Among all tested co-catalysts, 4-(dimethylamino)pyridine (DMAP) and bis(triphenylphosphine)iminium salts (X) proved to be the most useful. 
Most of them showed clearly higher catalytic activity and selectivity if they were used with a proper nucleophilic co-catalyst. Among the examples described till now, the complexes of Zn(II), Al(III), Fe(III), Cr(III), Co(III), and Mn(III) with ligands bearing N- and/or O-donor atoms, such as β-diketiminates, salens, porphyrinates, and some others polydentate ligand types, turned out to be particularly useful in the ROCOP of cyclic anhydrides and epoxides. Following this work, the increased attention has been paid by researchers to examine the metal complexes based on multidentate ligands. The authors were the first to show that perfectly alternating aliphatic polyesters with the high molecular weight and the narrow molecular weight distribution might also be obtained in the ROCOP reactions when an appropriate designed catalyst is applied. reported on the use of β-diketiminate zinc complexes as catalysts for the copolymerization of cyclic anhydrides with epoxides. The next noticeable progress in the development of catalytic systems for ROCOP occurred about twenty years later, in 2007, when Coates et al. The same authors also showed that Lewis bases used as nucleophilic co-catalysts, i.e., quaternary ammonium salts, improved sufficiently the catalytic activity of metal complexes (Lewis acids). The first significant progress in this field was done only in 1985, when Inoue and Aida published an article describing the use of single-site porphyrin aluminum complexes as catalysts for the copolymerization of phthalic anhydride and propylene oxide. The ROCOP reaction of cyclic anhydrides and epoxides has been known since the 1960s however, it did not attract much attention from researchers for many years, due to the difficulties in obtaining high-molecular-weight polyesters and eliminating the side reactions of epoxide polymerization. The resulting polyesters were characterized by M n up to 20.6 kg mol −1 and narrow dispersity, and they did not contain polyether units. When the molar ratio of ::: = 250:250:1:1 was applied, the most active complex (R 1-X = CH 2NMe 2, R 2 = F) allowed to copolymerize phthalic anhydride with differently substituted epoxides (cyclohexene oxide, 4-vinylcyclohexene oxide, styrene oxide, phenyl glycidyl ether, propylene oxide, butylene oxide, and epichlorohydrin) within 240 min at 110 ☌. The binary catalytic systems turned out to be more active than the complexes used individually, and DMAP proved to be the best choice as a co-catalyst. The co-catalyst was selected from the following organic bases: PPh 3, DMAP, 1-butylimidazole, or DBU. 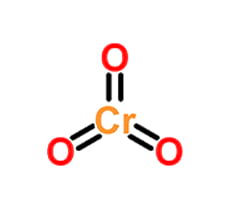
They were used individually or as a part of the binary catalytic systems with nucleophilic co-catalysts. Eight complexes differing in the donor-pendant group (R 1) and substituents (R 2) in phenolate units were examined as catalysts of the model reaction between phthalic anhydride and cyclohexane oxide in toluene. The effect of ligand structure on the catalytic activity of amine-bis(phenolate) chromium(III) complexes in the ring-opening copolymerization of phthalic anhydride and a series epoxides was studied.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
